Albert Virgili, Albert Granados, Carlos Jaime, Rosa Suárez-López, Teodor Parella and Eva Monteagudo
Cite this: J. Org. Chem. 2020, 85, 11, 7247–7257 https://doi.org/10.1021/acs.joc.0c00578

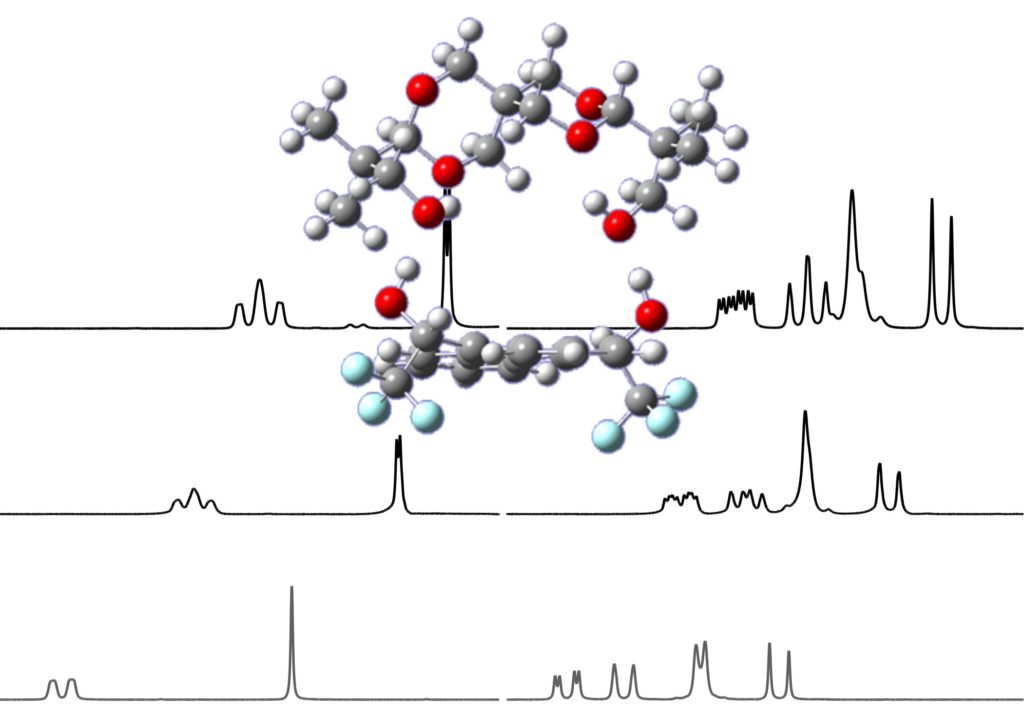
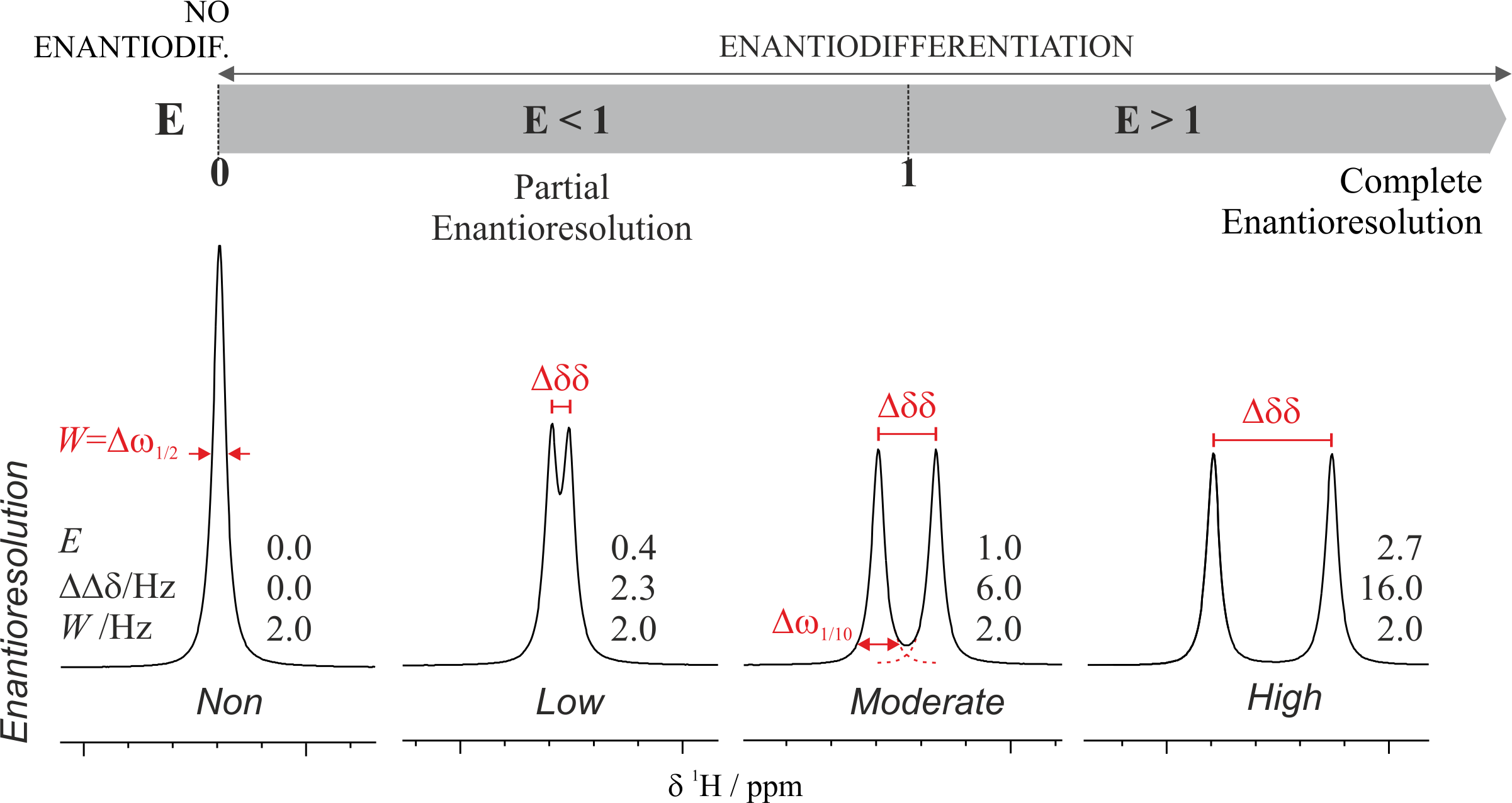
Herein, we perform for the first time a preliminary NMR and computational study of the spiroglycol structure. Spiroglycol is a highly symmetrical molecule, but it should be chiral due to the presence of a chiral axis. The presence of two enantiomers was demonstrated performing NMR enantiodifferentiation experiments using α,α′-bis(trifluoromethyl)-9,10-anthracenedimethanol (ABTE) as a chiral solvating agent (CSA). The addition of 0.6 equiv of ABTE allows the differentiation of several spiroglycol proton signals. The lack of resolution observed in the proton spectrum can be tackled through the corresponding 13C NMR spectrum where a significant enantiodifferentiation at the spirocarbon atom was observed. In order to physically separate both enantiomers, a SPG derivatization with camphorsulfonic acid and Mosher’s acid was performed affording the corresponding diastereoisomeric ester mixtures. Computations performed with the Gaussian16 package showed that the enantiodifferentiation is mainly due to the different compound thermodynamics stability.