“Molecule confirmation and structure characterization of pentatriacontatrienyl mycolate in Mycobacterium smegmatis” by M. Llorens-Fons, E. Julián, M. Luquin and M. Pérez-Trujillo. Chemistry and Physics of Lipids, 2018, Accepted Manuscript. DOI: https://doi.org/10.1016/j.chemphyslip.2017.12.006
“Molecule confirmation and structure characterization of pentatriacontatrienyl mycolate in Mycobacterium smegmatis” by M. Llorens-Fons, E. Julián, M. Luquin and M. Pérez-Trujillo. Chemistry and Physics of Lipids, 2018, Accepted Manuscript. DOI: https://doi.org/10.1016/j.chemphyslip.2017.12.006
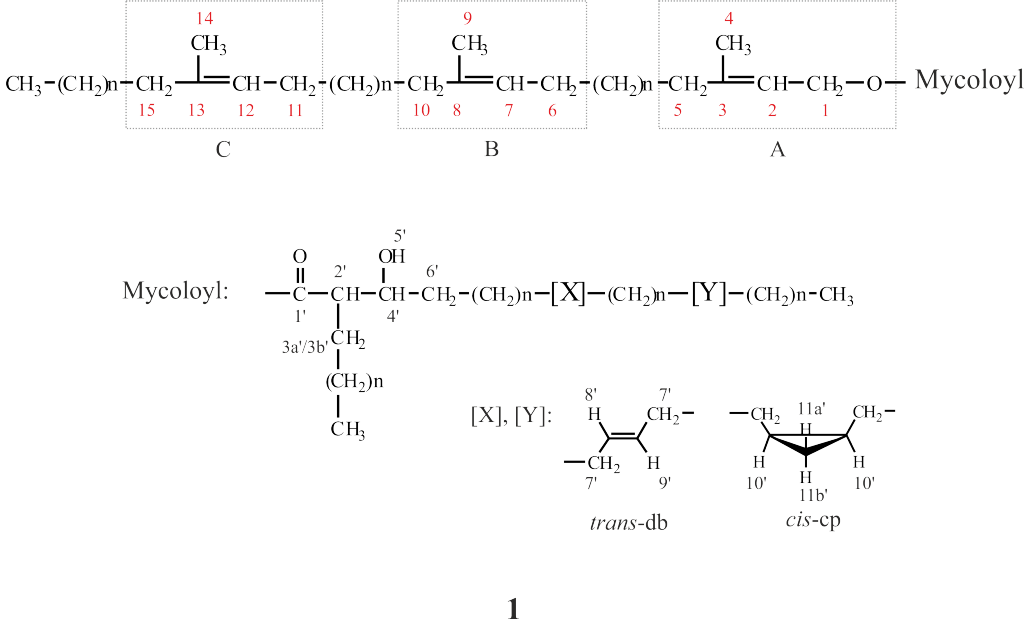
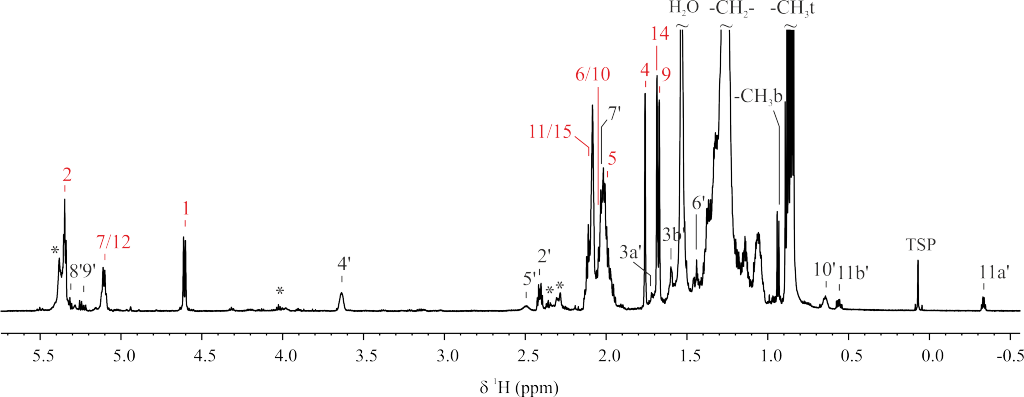
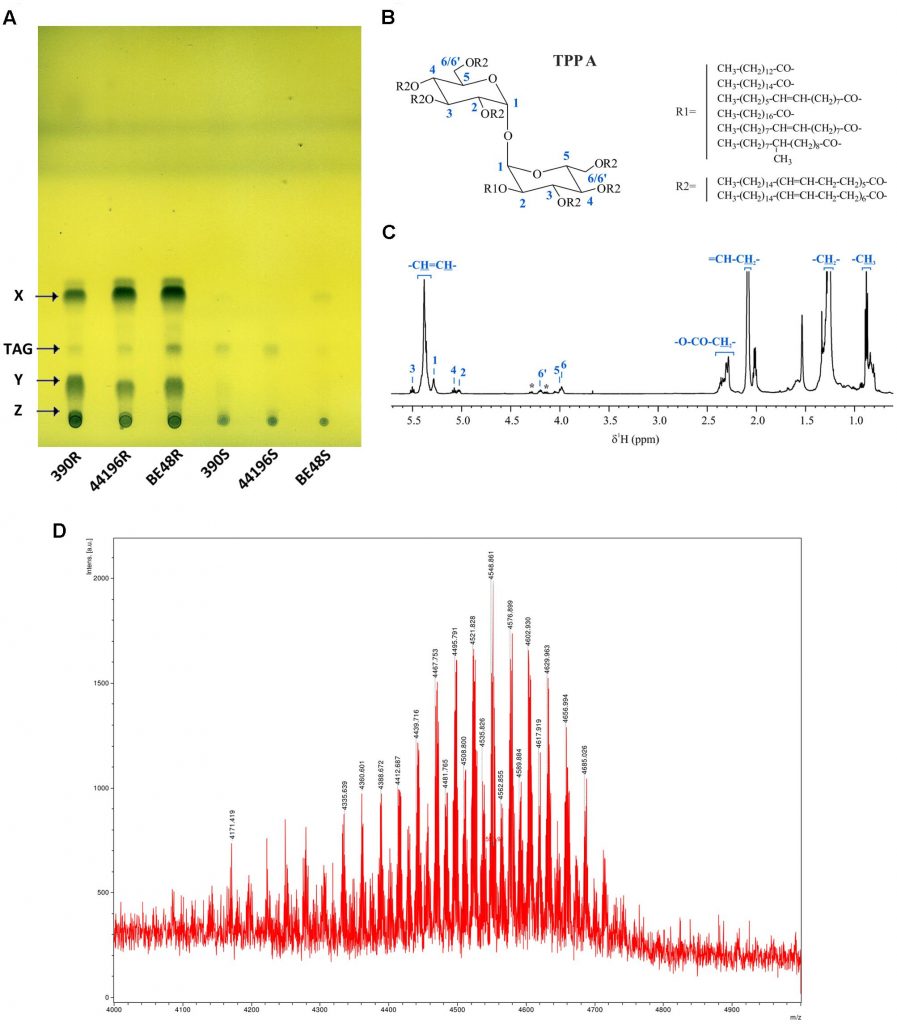
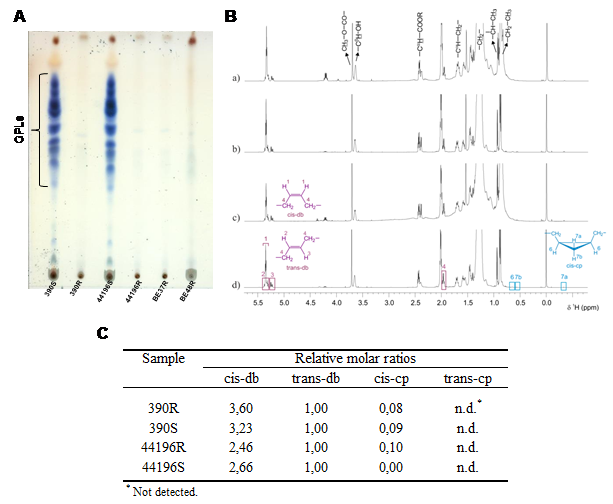
Mycobacterium smegmatis is often used to study the different components of mycobacterial cell wall. Mycolic acids are important components of mycobacterial cell wall that have been associated with virulence. Recently, a novel lipid containing mycolic acids has been described in M. smegmatis. However, some uncertainties regarding the structure of this molecule named mycolate ester wax have been reported. The objective of this work was to perform an in depth structural study of this molecule for its precise characterization. Using 1H and 13C NMR spectroscopy, the molecular structure of mycolate ester wax found in M. smegmatis has been elucidated. The characterization was complemented with MS analyses. This molecule is formed by a carbon chain with three methyl substituted olefinic units and a mycolate structure with trans double bonds and cis cyclopropane rings. The present molecular study will facilitate the detection and identification of pentatriacontatrienyl mycolate (PTTM) in future studies by the performance of a simple 1D 1H NMR experiment.