Job offer now closed and replaced by this job offer.
We are recruiting an Early Stage Researcher to work on the implementation of high-resolution MRSI methods in a pre-clinical scanner as part of the INSPiRE-MED European project.

We seek a highly motivated and qualified individual as Early Stage Researcher for a three-year applied research project. The successful candidate will contribute to the development of advanced biomedical research tools in the field of Magnetic Resonance Spectroscopy and Imaging, and its application to the clinical day-to-day practice.
Project description: This position is one of the 15 ESR positions of the INSPiRE-MED European Training Network, which focuses on the development of Magnetic Resonance Spectroscopy (MRS) and MR Spectroscopic Imaging (MRSI) combined with Positron Emission Tomography (PET), enhanced by machine learning techniques.
The main aim of the PhD project (ESR4) will be the implementation of innovative high spatial resolution MRSI methods in a pre-clinical scanner. The ultimate goal will be the validation of optimal methods for improving imaging biomarker development of brain tumour in longitudinal studies of therapy response in mouse glioblastoma models. The project will involve evaluation of the methodology performance limits, repeatability and reproducibility compared to stock Bruker Biospec MRSI sequences and the assessment of speed-up MRSI methods in a 7-Tesla pre-clinical scanner.
Continue reading Job offer for the INSPiRE-MED project

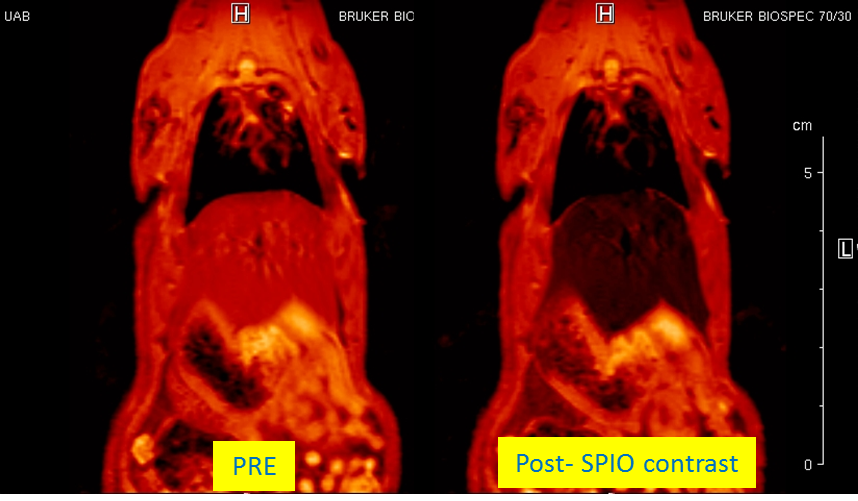

 “Assessment of biodistribution using mesenchymal stromal cells: Algorithm for study design and challenges in detection methodologies” by Reyes B, Coca MI, Codinach M, López-Lucas MD, Del Mazo-Barbara A, Caminal M, Oliver-Vila I, Cabañas V,
“Assessment of biodistribution using mesenchymal stromal cells: Algorithm for study design and challenges in detection methodologies” by Reyes B, Coca MI, Codinach M, López-Lucas MD, Del Mazo-Barbara A, Caminal M, Oliver-Vila I, Cabañas V, 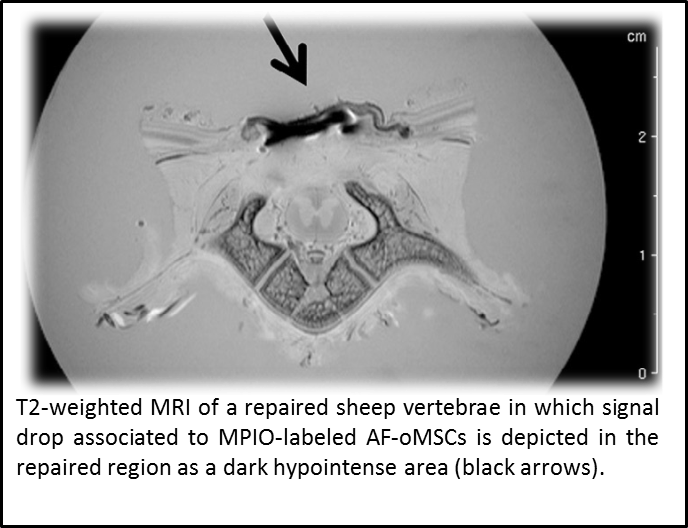
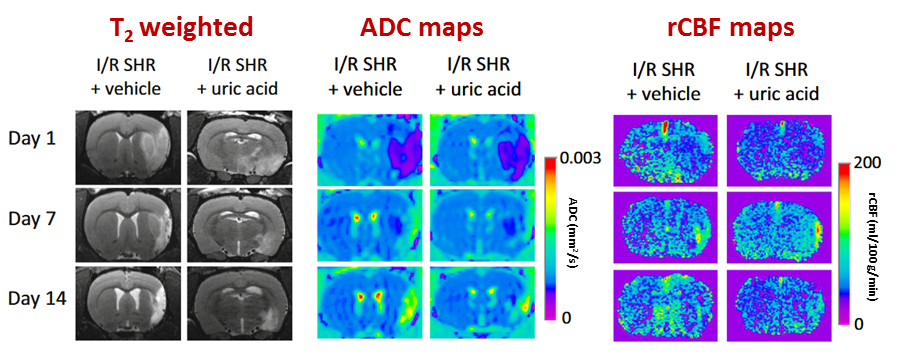
 “Metronomic treatment in immunocompetent preclinical GL261 glioblastoma: effects of cyclophosphamide and temozolomide” by by L. Ferrer-Font, N. Arias-Ramos,
“Metronomic treatment in immunocompetent preclinical GL261 glioblastoma: effects of cyclophosphamide and temozolomide” by by L. Ferrer-Font, N. Arias-Ramos, 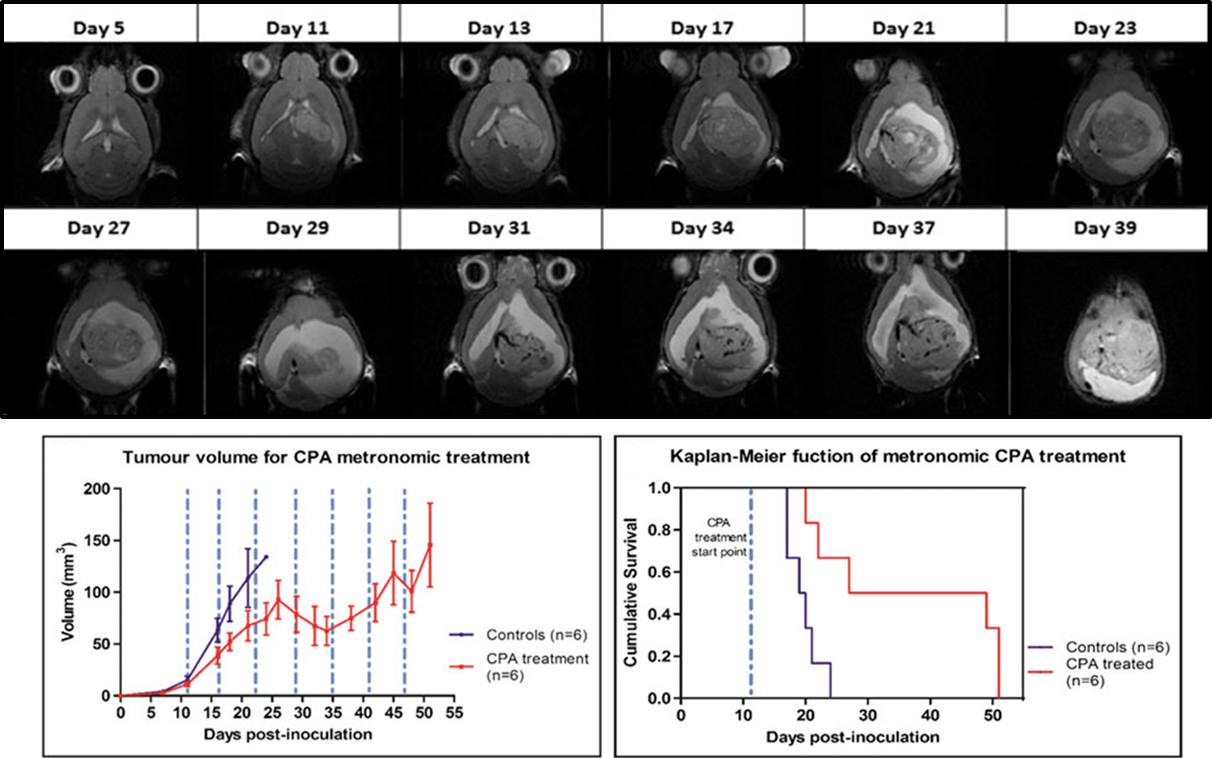

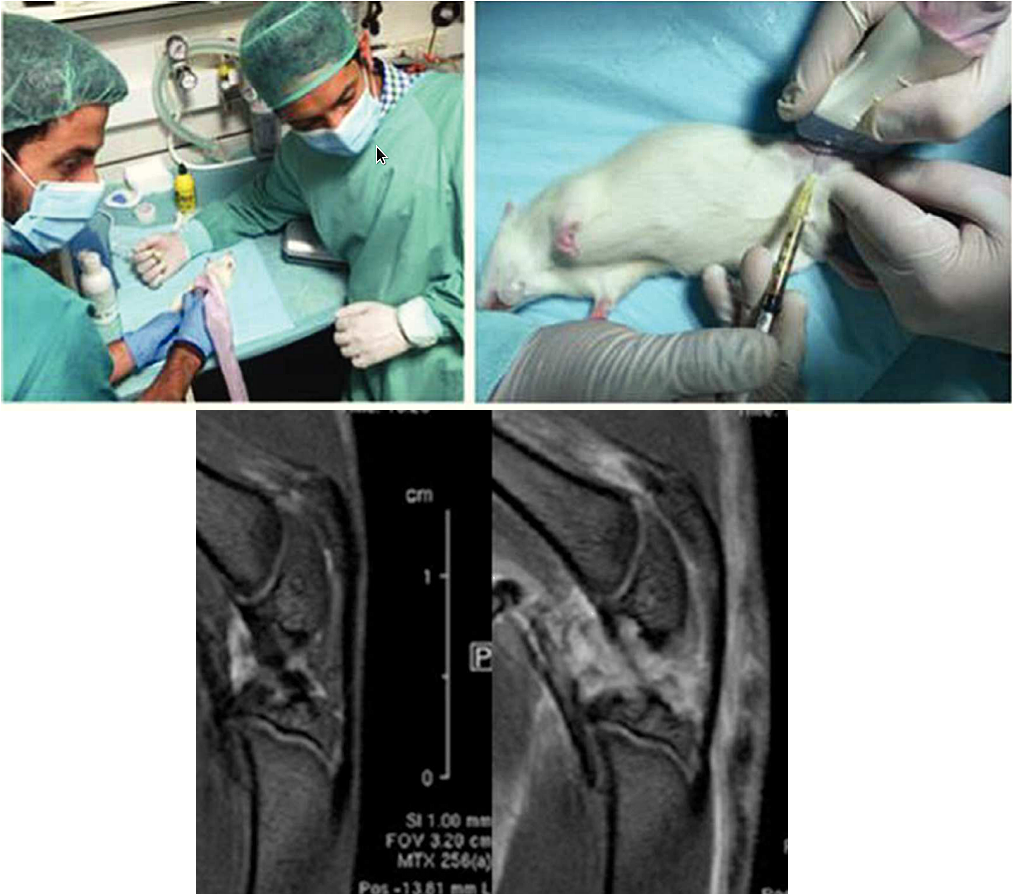
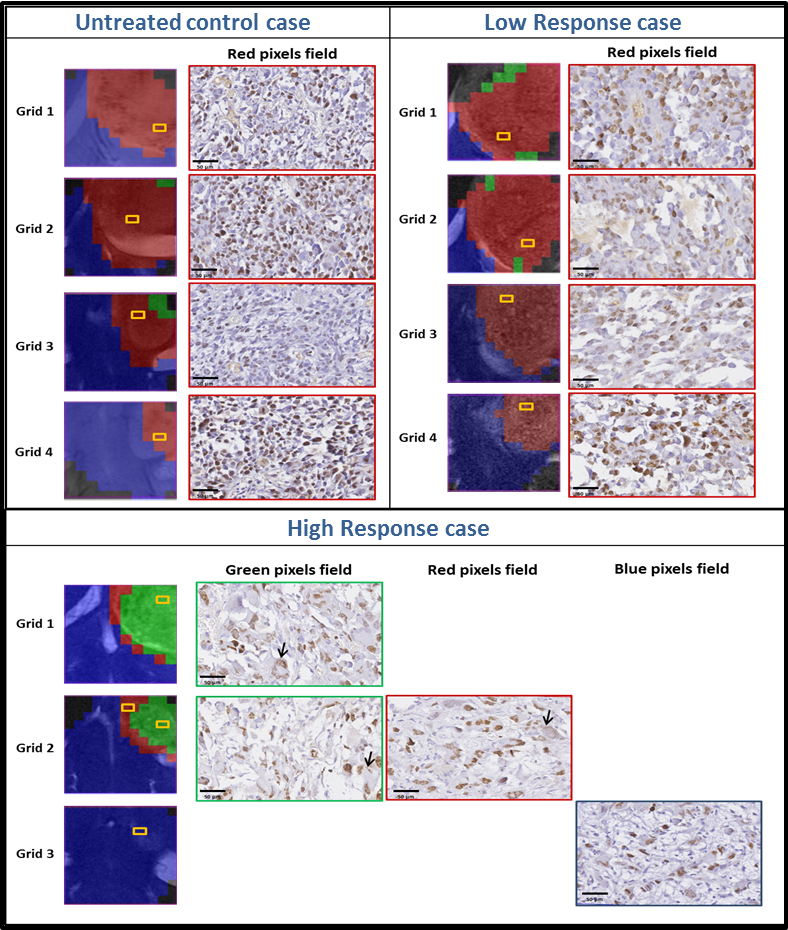
 “Metabolomics of Therapy Response in Preclinical Glioblastoma: A Multi-Slice MRSI-Based Volumetric Analysis for Noninvasive Assessment of Temozolomide Treatment” by N. Arias-Ramos, L. Ferrer-Font,
“Metabolomics of Therapy Response in Preclinical Glioblastoma: A Multi-Slice MRSI-Based Volumetric Analysis for Noninvasive Assessment of Temozolomide Treatment” by N. Arias-Ramos, L. Ferrer-Font, 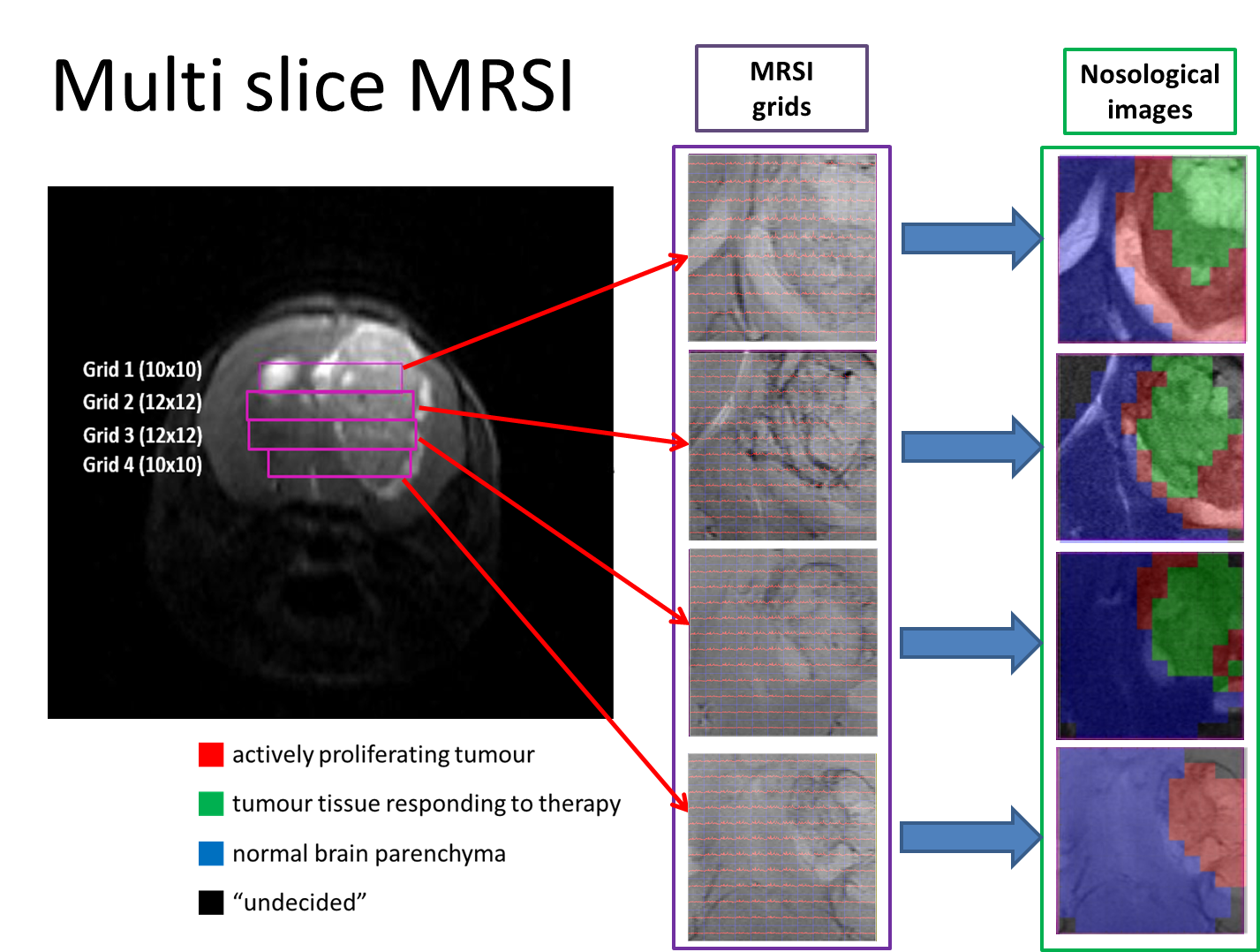 Therefore, the goal of this work was to acquire 3D-like information from preclinical GBM under a longitudinal treatment protocol, using a multi-slice MRSI approach.
Therefore, the goal of this work was to acquire 3D-like information from preclinical GBM under a longitudinal treatment protocol, using a multi-slice MRSI approach.