On 21st March 2018 I sucessfully defended my PhD Thesis entitled: “New Applications of Covariance NMR and Experimental Development for Measurements of Homonuclear Coupling Constants in Overlapping Signals” (ISBN: 9788449079252), supervised by Dr. Teodor Parella and Dr. Pau Nolis, and obtained the degree of Ph.D. in Chemistry at the Department of Chemistry, Universitat Autònoma de Barcelona.
The thesis can be downloaded in PDF format from the TDX repository and from the TESEO repository.
Abstract
The experimental results obtained in this thesis are presented in the form of three papers published in NMR specialised scientific peer-reviewed journals.
- Exploring the use of Generalized Indirect Covariance to Reconstruct Pure shift NMR Spectra: Current Pros and Cons. André Fredi, Pau Nolis, Carlos Cobas, Gary E. Martin and Teodor Parella. Journal of Magnetic Resonance, Volume 266, May 2016, Pages 16-22. DOI: 10.1016/j.jmr.2016.03.003
- Access to experimentally infeasible spectra by pure-shift NMR covariance. André Fredi, Pau Nolis, Carlos Cobas and Teodor Parella. Journal of Magnetic Resonance, Volume 270, September 2016, Pages 161-168. DOI: 10.1016/j.jmr.2016.07.010
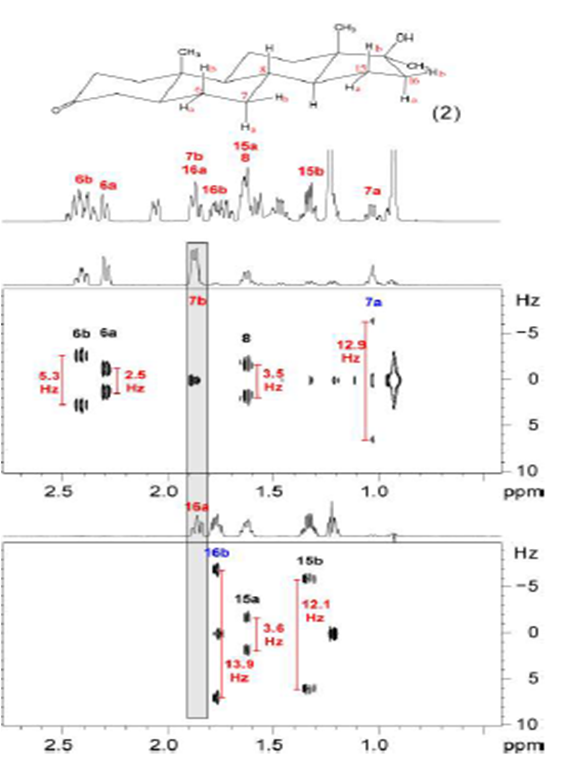
- Accurate measurement of JHH in overlapped signals by a TOCSY‐edited SERF Experiment. André Fredi, Pau Nolis and Teodor Parella. Magnetic Resonance in Chemistry, Volume 55, Issue 6, June 2017, Pages 525-529. DOI: 10.1002/mrc.4572
The first two articles deal with the use of covariance NMR as a general method to generate novel psNMR spectra. The last work describes a new selTOCSY G-SERF experiment, for accurately measuring JHH in overlapped regions.
The first publication describes a novel general protocol to generate psNMR spectra by Covariance NMR. This new approach is unique in NMR spectroscopy; giving a cheap, fast an easy way to reconstruct psNMR spectra without spending time in the spectrometer. This new strategy has been referenced to as psNMR Covariance.
The concept of psNMR Covariance has been extended in the second publication by inserting Multiplicity-Edited (ME) information into 2D experiments that are difficult or even impossible to achieve experimentally. It is shown how the ME information can be efficiently transferred to a set of homonuclear and heteronuclear 2D NMR spectra by Covariance processing, reconstructing new psME spectra in a fast way. Finally, G-SERF and related methods only work for isolated 1H signals on which selective excitation can be successfully applied.
Unfortunately, as it happens in other frequency-selective experiments, this approach fails for overlapped signals. A doubly-selective TOCSY G-SERF scheme is presented in the third publication to circumvent this limitation, by measuring JHH efficiently even for protons resonating in crowded regions.




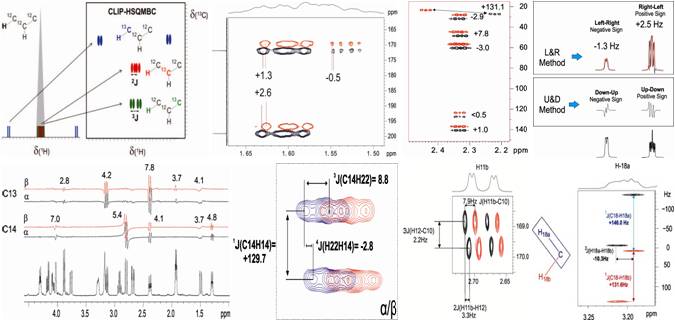
 “Simultaneous measurement of J(HH) and two different nJ(CH) coupling constants from a single multiply-edited 2D cross-peak”, by Josep Saurí and
“Simultaneous measurement of J(HH) and two different nJ(CH) coupling constants from a single multiply-edited 2D cross-peak”, by Josep Saurí and